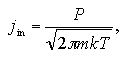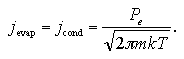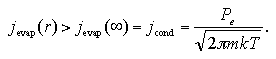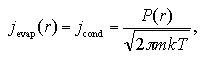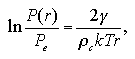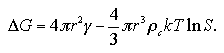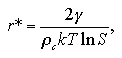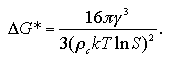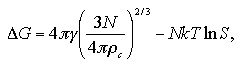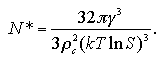Chapter 1:
INTRODUCTION
|
|
Supersaturated vapor generates various sizes of nuclei. If the generated nucleus was smaller than a certain size, the nucleus should collapse and disappear. On the other hand, the larger nucleus should grow up to a liquid droplet. The size and the nucleation free energy of the critical nucleus play important roles for growth of the liquid droplet. Experimental and theoretical studies for the homogeneous nucleation have been much widely carried out especially about the critical nucleus.
|
|
1.1. Equilibrium Theory of Phase Transition (omitted) |
|
1.2. Kinetics of the Homogeneous Nucleation |
|
According to the phase equilibrium theory, when the system is superheated or supercooled, the nuclei appear and grow immediately in the unstable phase, and the system ought to transfer toward the more stable phase. However, a metastable phase, a few superheated or supercooled systems, exists actually in which the nucleation and the phase transition do not occur. While, if the superheating or the supercooling ratio is above the certain criticality, the nucleation and the phase transition are observed. Such phenomenon is understood with kinetics of the nucleus growth. |
1.2.1. Gibbs-Thomson's formula for the equilibrium pressure of the cluster
In the supersaturated vapor, the molecules collide each other and the nuclei grow up orderly as a monomer, dimer, trimer, tetramer... and larger cluster. In opposite, the going away of the molecule(s) from the cluster also occurs simultaneously. The nucleation rate J is expressed as the difference between the growth rate Jcond and the decay rate Jevap:
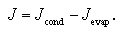 | (1.4) |
Here, we consider about the nucleation rate in comparison with the kinetics of the bulk liquid-vapor equilibrium system.
The number of the vapor phase molecules that enter into the liquid phase per unit time per unit area, jin, is described from the kinetics of vapor molecules:
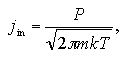 | (1.5) |
where m is mass of the molecule, k is the Boltzmann constant, and P is the system pressure. When the entered vapor molecules can be assumed to condense immediately into the liquid phase, the jin can be regarded as the frequency of the condensation per unit area jcond. In the liquid-vapor equilibrium system, the jcond at the equilibrium pressure Pe equals to the frequency of the evaporation per unit time per unit area, jevap, as
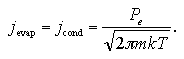 | (1.6) |
This corresponds to the equilibrium theory in which the chemical potentials of the both phases are identical if the system is in equilibrium as μliquid = μvapor.
On the other hand, in the case of the small-size cluster in the (supersaturated) vapor phase, the contribution of the surface excess energy becomes remarkable because the ratio of the surface area against the volume of the cluster becomes larger, while the jevap gets a dependence on the radius of cluster r; where the cluster is assumed as spherical-shape for easy consideration. The evaporation frequency of the cluster is larger than the one of the bulk liquid phase as
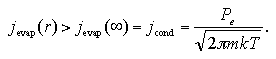 | (1.7) |
Here, jevap(r) is the decay frequency of the cluster that depends on r, while jevap(∞) is the one of the bulk liquid that can be regarded as r = ∞, because the surface is a plane. When the system pressure rises as far as obtain an identical decay-growth frequency,
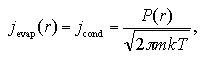 | (1.8) |
now!! now!! now!!
the cluster equilibrates in the supersaturated vapor phase. The symbol P(r) is the pressure of the supersaturated vapor phase that equilibrates with the cluster with the radius of r. Relation between the P(r) and Pe is given as the Gibbs-Thomson's formula:
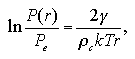 | (1.9) |
where ρc is the number density of the cluster and γ the density of the surface excess energy per unit area. If the system pressure is more than the equilibrium pressure of the bulk phases while less than the one of the small-size cluster, P(r) > P > Pe, the cluster decays as soon as which nucleation and the vapor preserves as the supersaturated phase. In other words, P(r)/Pe becomes larger when the r becomes smaller, then the P(r) is more than the system pressure, and there is strong probability that the cluster should decay without growing. On the other hand, the larger cluster which P(r) is less then the system pressure should grow probably. Between both the sizes, the cluster with a certain radius of r* obtains an identical pressure as P(r*) = P, and equilibrates in the vapor phase. The r* is termed as the critical radius, and the cluster with the radius of r* as the critical nucleus. In the spontaneous formation of the various sizes of nuclei due to density fluctuation of the supersaturated vapor phase, the formation frequency of the cluster with the radius of r > r* per unit volume per unit time relates the nucleation rate J. The nucleation rate is interested in which dependence on the supersaturation ratio. The summary of the Gibbs-Thomson's formula is shown in Fig. 1.2.
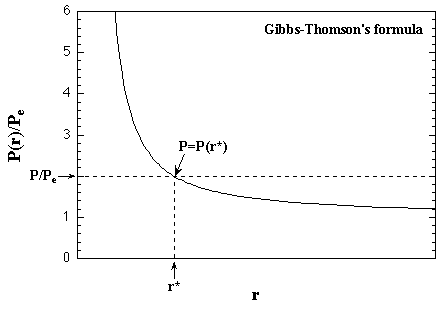 |
| Fig. 1.2. The relation of the equilibrium pressure of the cluster with the radius of r and the one of the bulk vapor phase derived by the Gibbs-Thomson's formula. When the radius of cluster is a critical value r*, the equilibrium pressure of the cluster is equivalent to the system pressure; the critical radius is given as r* = 2γ/ρckTln(P/Pe), from eq. 1.9. The broken lines show a case of P = 2Pe. |
|
1.2.2. Classical nucleation theory
In this section, we consider about the free energy change of the system accompanying the homogeneous nucleation. The classical nucleation theory considers the nucleation free energy ΔG as
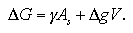 | (1.10) |
Here, the first term indicates the surface contribution; γ is surface excess energy per unit area and As surface area of the cluster. While the second term is the bulk contribution; Δg is free energy change per unit volume between the bulk liquid phase and bulk the vapor phase, and V volume of the cluster. When the shape of cluster is assumed as spherical, As and V are expressed by the radius of the cluster r as As = 4πr2 and V = (4/3)πr3. The free energy change Δg is obtained as Δg = -ρckTlnS from assumption of the vapor phase as an ideal gas, where ρc is density of the cluster and S supersaturation ratio. Such relations rearrange the eq. 1.10 as
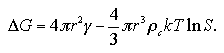 | (1.11) |
The radius r* and the free energy ΔG* of the critical nucleus are obtained as
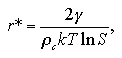 | (1.12) |
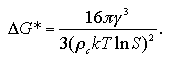 | (1.13) |
The supersaturation ratio is expressed as S = (P/Pe), where P and Pe are the pressures of the system and the equilibrated vapor phase. Accordingly, the expression of the critical radius of eq. 1.12, corresponds to the Gibbs-Thomson's formula shown in eq. 1.9. The r* is in inverse proportion to the lnS, therefore the larger supersaturation makes the r* smaller.
Equation 1.11 is rearranged as a function of the number of cluster molecules by the relation of N = ρcV:
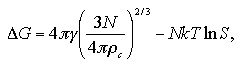 | (1.14) |
and the size of the critical nucleus N* is obtained as
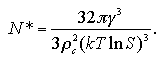 | (1.15) |
Figure 1.3 shows the nucleation free energy ΔG from the classical theory.
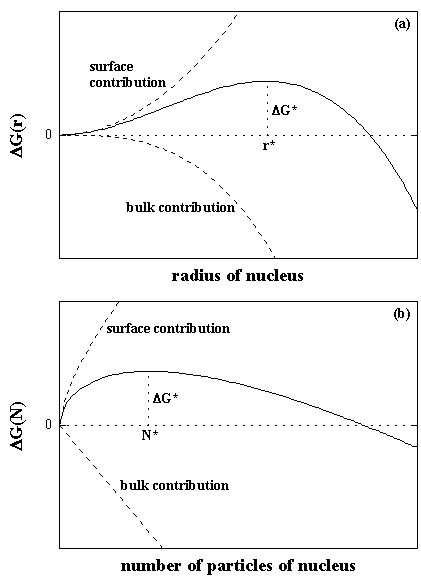 |
| Fig. 1.3. Nucleation free energy ΔG as a function of (a) the cluster radius r and (b) the number of cluster molecules N predicted by the classical nucleation theory. The classical nucleation theory explains the ΔG as the sum of the surface contribution and the bulk contribution. Due to density fluctuation in the supersaturated vapor phase, if the generated cluster grows larger than the critical nucleus, the cluster should grow to macroscopic liquid droplet. |
|
1.2.3. Nucleation rate (omitted)
|
|
1.3. Purpose of This Study |
The studies for the homogeneous nucleation with computer simulations have been widely performed. Giving the studies of recent years, Yasuoka and Matsumoto carried out large-system ensemble molecular dynamics (MD) simulations and estimated the size of critical nucleus and the free energy of nucleation from the cluster size distribution.[2-4] Oh and Zeng reported cluster size dependence of the nucleation energy by constrained Monte Carlo (MC) simulations in which applied upper limit of the cluster size.[5] They also performed small-system grand canonical ensemble MC method in which the cluster-vapor interaction is effectively taken into account.[6] Wolde and Frenkel obtained the nucleation energy by umbrella sampling.[7] Kusaka and Oxtoby suggested an approach to cluster simulation without a cluster criterion that is determination particle by particle whether it belongs to cluster or vapor phase for instantaneous cofiguration.[8]
The previous computational works performed on some particular states, then that do not predict the nucleation free energy on a wide range of the temperature and the pressure. In this work, we suggest a simple model and a method for the purpose of obtaining the free energy of the homogeneous nucleation by using the canonical MC simulations. In which, we assumed a technique for direct estimation of the free energy of the system, and we obtained the nucleation free energy as difference between the free energies of the cluster and the supersaturated vapor phase. Moreover, we approximated an equation of state (EOS) for the Helmholtz free energy of the nucleation from the MC results. We performed the two series of MC simulations; under a fixed volume per particles and various volumes per particle. In the former, we estimated the EOS as a function of the number of particles and temperature, while in the latter, we included the volume dependence toward the EOS. On the basis of the EOS in canonical ensemble, we rearranged the EOS to estimate Gibbs free energy of the nucleation as a function of the number of particle, the temperature, and the pressure.
|