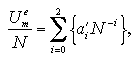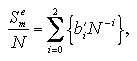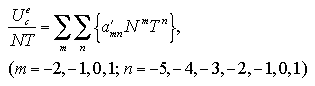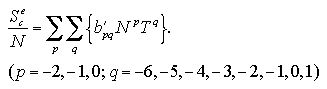| <- back | INDEX | next -> |
|
Chapter 3: MONTE CARLO SIMULATIONS UNDER A FIXED VOLUME PER PARTICLE | ||||||||
| 3.1. Monte Carlo Results | ||||||||
|
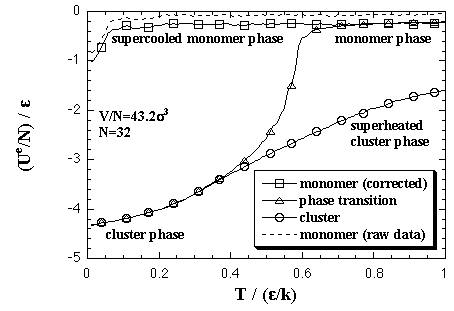
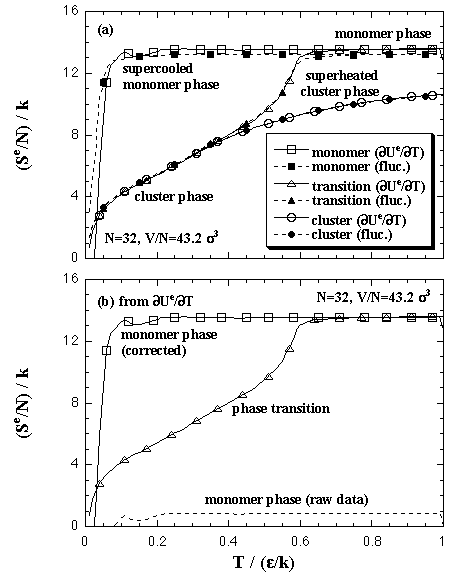
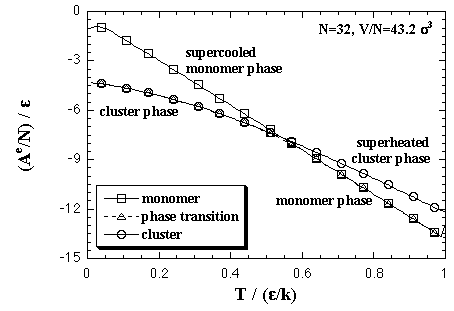
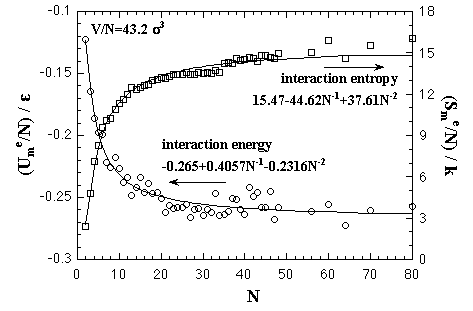
First, we performed the MC simulations under a fixed volume per particle to estimate the EOS of the Helmholtz free energy of the homogeneous nucleation as a function of the number of particles and the temperature. The volume per particle V/N = 43.2 σ3. This value corresponds to supersaturated vapor phase and recent MD simulations by Yasuoka and Matsumoto.[2,3] Here we explain and discuss about the results of the MC simulations. Both the interaction energy and the interaction entropy increase in proportion to the number of particles in a sufficiently large system. Then, we mainly treat about the thermodynamic quantities as values per particle. As an example of MC results, the thermodynamic properties of a 32-particle system are shown below. Figure 3.1 shows averaged interaction energy per particle versus temperature. In the phase transition calculation, the stabilized cluster decomposes to the monomer phase at a certain temperature that depends on the number of particles. The phase transition temperature is ca. 0.57 ε/k in the 32-particle system. Though the equilibrated value of the decomposed monomer phase in the phase transition calculation should correspond to the value of the monomer phase calculation, they did not agree. As we stated above, the each interparticle distance is restricted to be longer than 1.5 σ in the monomer phase calculation, yet the restriction is not applied in the phase transition calculation. From this difference of condition, it is conjectured that the equilibrated interaction energy of the two calculations would disagree. The monomer phase calculation gives an ideal vapor phase; however, we consider that the decomposed monomer phase in the phase transition system is more close to the real monomer phase. Accordingly, we corrected the curve of the monomer phase calculation to agree with the equilibrated value of the decomposed monomer phase in the phase transition calculation. Moreover, this correction aims to create a relation between the cluster phase calculation and the monomer phase calculation. eq. 2.7 is shown in Fig. 3.3. Here, the corrections for the interaction energy were also adopted for the curves of monomer phase calculation as the figure (b). In the figure (a), we confirmed the equality of the interaction entropy from the two ways of the heat capacity estimation (eqs. 2.5 and 2.6). Accordingly, we decided to adopt the numerical differentiation of the interaction energy, eq. 2.5, for the heat capacity estimation. Fig. 3.4. The intersection of the curves of the cluster phase and the monomer phase calculations indicates a phase transition temperature, it is 0.55 ε/k at the 32-particle system. On the phase transition calculation, the cluster decomposes gradually over some range of temperature that is centering on the phase transition temperature since the calculation was not adopted any restrictions on the configuration. Accordingly, a peak of the heat capacity on the phase transition calculation, 0.57 ε/k in the 32-particle system, does not always agree with the intersection of the curves of the monomer phase and the cluster phase calculations. The phase transition temperatures from the peak of the heat capacity and the intersection are compared in Fig. 3.5 (omitted). There is not much different in both plots.
| ||||||||
| 3.2. Approximated Functions for the Equation of State | ||||||||
The interaction energy Ue, the interaction entropy Se, the interaction term of Helmholtz free energy Ae, and the nucleation free energy change ΔA can be described as a function of the number of particles and the temperature. We assumed the approximated functions for development of an equation of state (EOS) of the nucleation free energy. Which are designed as the power series for easy handling:[13,14]
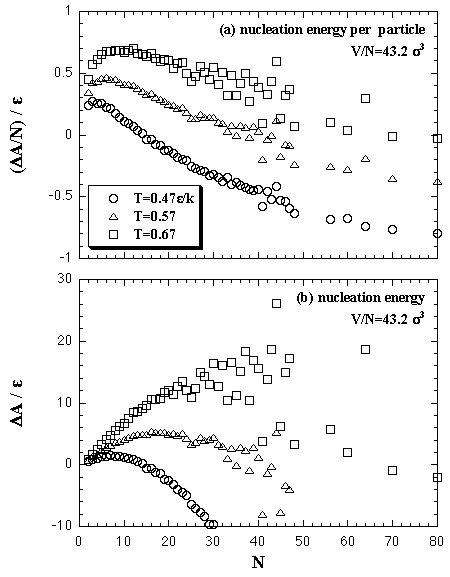
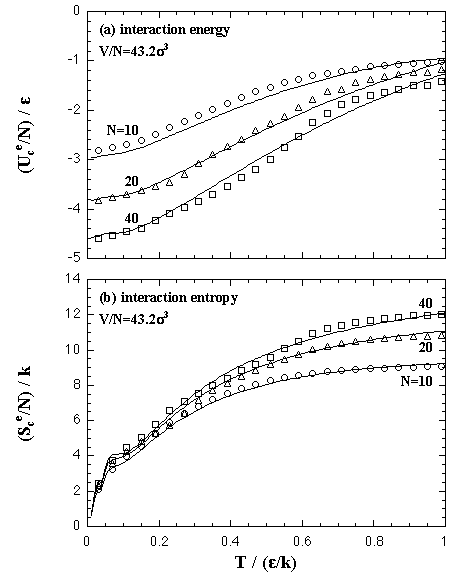
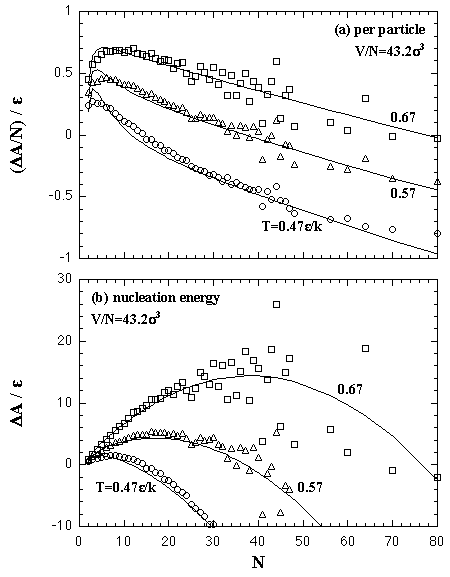
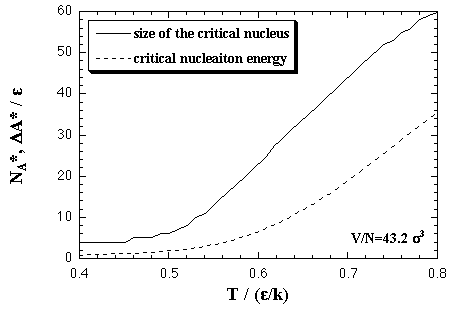
The interaction entropy is obtained from the interaction energy by eqs. 2.4-2.8. Therefore, in essential, the function for the interaction energy ought to predict not only the interaction energy but also the interaction entropy, and two approximated functions are sufficient concerning the EOS: for the monomer phase and for the cluster phase. In the case of the monomer phase, we assumed that the interaction energy and the interaction entropy have no dependence on the temperature, then the interaction terms of the heat capacity and the entropy should become zero. Accordingly, the function for the interaction entropy is described separately from the one for the interaction energy, and we used the equilibrated value of the decomposed monomer phase in the phase transition calculation instead of the value of the monomer phase calculation. On the other hand, the interaction energy and the interaction entropy of the cluster phase were approximated unsuccessfully by the one expression for the interaction energy. It may be caused by the error from the limited number of MC steps and the dispersion of the MC results that is due to the treating of the meta- or unstable phase: the superheated cluster. The coefficients of the approximated functions, a'i, b'i, a'mn, and b'pq are determined by least-squares fittings. The fitting algorithm is the Modified Gram-Schmidt method. Figures 3.7-3.10 show the results of the least-squares fittings. In the figures, the circles, triangles, and squares indicate the MC results, and the solid curves are the approximated functions. It can be seen that the functions well reproduce the MC results. The curve of the nucleation free energy, like the Fig. 3.10 (b), has a maximum against the number of particles that corresponds to the values of the critical nucleus. Which, the free energy ΔA* and the size NA*, are shown in Fig. 3.11. Both curves simply increase against the temperature. |
| <- back | INDEX | next -> |